Difference between revisions of "YGR079W"
SGDwikiBot (talk | contribs) (Automated import of articles) |
(→Community Commentary) |
||
| (31 intermediate revisions by 2 users not shown) | |||
| Line 4: | Line 4: | ||
{|{{Prettytable}} align = 'right' width = '200px' | {|{{Prettytable}} align = 'right' width = '200px' | ||
|- | |- | ||
| − | |valign="top" nowrap bgcolor="{{SGDblue}}"| '''Systematic name''' || [http:// | + | |valign="top" nowrap bgcolor="{{SGDblue}}"| '''Systematic name''' || [http://www.yeastgenome.org/cgi-bin/locus.pl?dbid=S000003311 YGR079W] |
|- | |- | ||
|valign="top" nowrap bgcolor="{{SGDblue}}"| '''Gene name''' ||'' '' | |valign="top" nowrap bgcolor="{{SGDblue}}"| '''Gene name''' ||'' '' | ||
| Line 13: | Line 13: | ||
|- | |- | ||
|valign="top" nowrap bgcolor="{{SGDblue}}"| '''Coordinates''' | |valign="top" nowrap bgcolor="{{SGDblue}}"| '''Coordinates''' | ||
| − | |nowrap| Chr VII: | + | |nowrap| Chr VII:640720..641832 |
| + | |- | ||
| + | |valign="top" nowrap bgcolor="{{SGDblue}}"| '''Primary SGDID''' || S000003311 | ||
|} | |} | ||
<br> | <br> | ||
| − | '''Description of | + | '''Description of YGR079W:''' Putative protein of unknown function; YGR079W is not an essential gene<ref name='S000071347'>Giaever G, et al. (2002) Functional profiling of the Saccharomyces cerevisiae genome. Nature 418(6896):387-91 |
{{SGDpaper|S000071347}} PMID 12140549</ref> | {{SGDpaper|S000071347}} PMID 12140549</ref> | ||
<br> | <br> | ||
| Line 37: | Line 39: | ||
{{ShortCenteredHR}} | {{ShortCenteredHR}} | ||
| − | + | {{UW-Stout}} | |
<!-- PLEASE ADD Community Commentary ABOVE THIS MESSAGE. See below for an example of community annotation --> | <!-- PLEASE ADD Community Commentary ABOVE THIS MESSAGE. See below for an example of community annotation --> | ||
| Line 45: | Line 47: | ||
J Biol Chem 278(5):3265-74</ref> | J Biol Chem 278(5):3265-74</ref> | ||
--> | --> | ||
| + | ===[[UW Stout/D2O SP22]]=== | ||
| + | [[Image:4.28.2022 1.png]] | ||
| + | |||
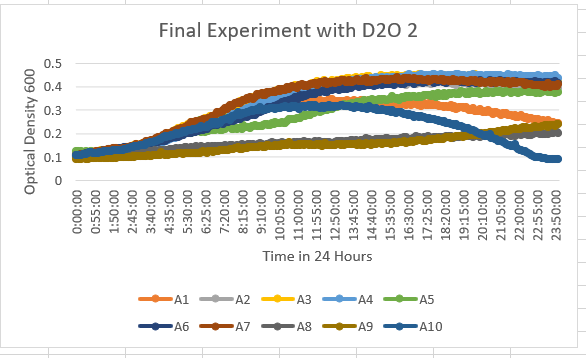
| + | The Knock Out Yeast Strain YGR079W (A8) Is more of a resistant strain to the 35% dilution of D20. With the range of maximum OD being about 0.2-0.3 OD 600. However, the growth of this is also a slower growth rate. | ||
| + | |||
| + | ===[[UW Stout/Sucrose Fermentation SP22]]=== | ||
| + | |||
| + | {| border="1" cellspacing="4" cellpadding="2" | ||
| + | |Gene | ||
| + | |Glucose | ||
| + | |Fructose | ||
| + | |Ethanol | ||
| + | |- | ||
| + | |Standard solution | ||
| + | |2.0000 | ||
| + | |0.2000 | ||
| + | |2.0000 | ||
| + | |- | ||
| + | |YDl109C | ||
| + | |0.3800 | ||
| + | |0.3933 | ||
| + | |0.3430 | ||
| + | |- | ||
| + | |YGL140C | ||
| + | |0.2212 | ||
| + | |0.2685 | ||
| + | |0.1867 | ||
| + | |- | ||
| + | |YOR111W | ||
| + | |0.3332 | ||
| + | |0.3598 | ||
| + | |0.1343 | ||
| + | |- | ||
| + | |YHL029C | ||
| + | |0.3870 | ||
| + | |0.2368 | ||
| + | |0.1151 | ||
| + | |- | ||
| + | |YDR307W | ||
| + | |0.4366 | ||
| + | |0.2487 | ||
| + | |0.0606 | ||
| + | |- | ||
| + | |YNL058C | ||
| + | |0.2710 | ||
| + | |0.3056 | ||
| + | |0.1577 | ||
| + | |- | ||
| + | |YCL049C | ||
| + | |0.4078 | ||
| + | |0.3052 | ||
| + | |0.1969 | ||
| + | |- | ||
| + | |YGR079W | ||
| + | |0.4042 | ||
| + | |0.1589 | ||
| + | |0.0080 | ||
| + | |- | ||
| + | |YBL113C | ||
| + | |0.3498 | ||
| + | |0.2012 | ||
| + | |0.1434 | ||
| + | |- | ||
| + | |BY4735 | ||
| + | |0.3171 | ||
| + | |0.3084 | ||
| + | |0.3541 | ||
| + | |} | ||
| + | |||
| + | ====Interpretation==== | ||
| + | This gene produced 2.26% of the amount of ethanol that the wild type produced. There is a high probability that this gene affects fermentation since there is such a low amount of ethanol produced but further tests are required. | ||
<protect> | <protect> | ||
| + | |||
| + | ===[[UW-Stout/UV Light SP22]]=== | ||
| + | |||
| + | As part of the University of Wisconsin Stout [[UW-STOUT/About|Orphan Gene Project]] this gene was tested under a UV Light [[UW-Stout/UV Light SP22|using this protocol.]] | ||
| + | |||
| + | '''RESULTS''' | ||
| + | |||
| + | [[File:ASHTONMIKOLOSKIFinal Project Graph.png|450px|]] | ||
| + | [[File:AshtonGene8.jpg|350px|]] | ||
| + | |||
| + | |||
| + | '''INTERPERTATION''' | ||
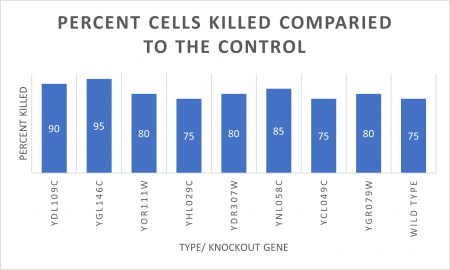
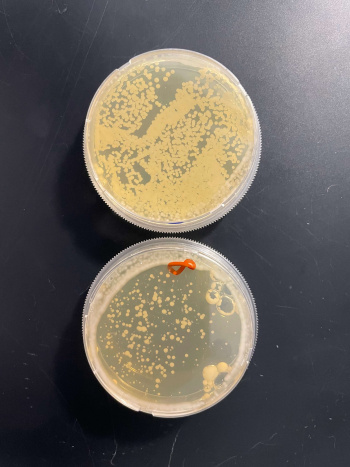
| + | In the graph and photos above, exposing this gene to 600 seconds of 400 Watt UV Light killed approximately 80% of yeast cell cultures, compared to its control counterpart, which was the same gene and amount of cells, just was not exposed to UV Light. | ||
| + | |||
| + | ===[[UW-Stout/Heat Shock SP22]]=== | ||
| + | |||
| + | As part of the University of Wisconsin Stout [[UW-Stout/About|Orphan Gene Project]] this gene was tested by exposing the cells to heat shock. | ||
| + | |||
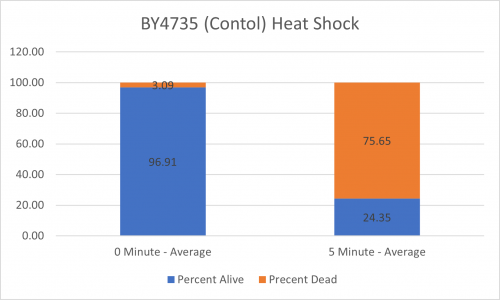
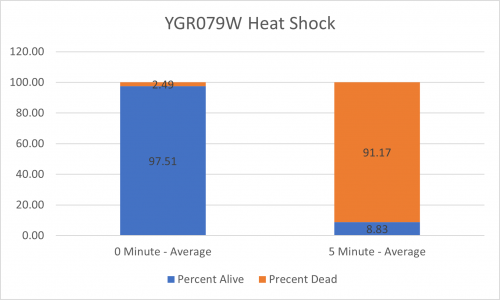
| + | ====Results==== | ||
| + | [[Image:BY4735 Control.png|BY4735 Control]] [[Image:YGR079W.png|YGR079W]] | ||
| + | [[Image:BY4735 Control Graph.png|BY4735 Control Graph|500px]] [[Image:YGR079W Graph.png|YGR079W Graph|500px]] | ||
| + | |||
| + | ====Interpretation==== | ||
| + | |||
| + | From the data gathered, there was a medium negative effect to knocking out this gene when it came to the yeast's ability to hold up to heat shock. The modified yeast cells were only 36% as resistant to heat shock than the control. | ||
| + | |||
| + | ===[[UW-Stout/Caffeine SP22]]=== | ||
| + | |||
| + | As part of the University of Wisconsin Stout [[UW-Stout/About|Orphan Gene Project]] this gene was tested by exposing the cells to 4mM of caffeine [[UW-Stout/Caffeine SP22|following this protocol]]. | ||
| + | |||
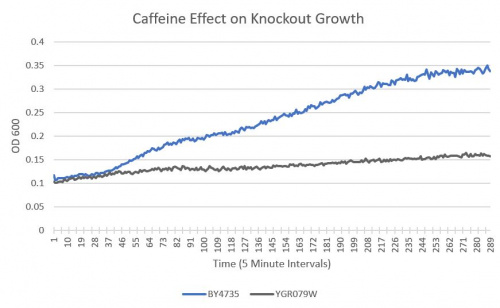
| + | ====Results==== | ||
| + | :BY4735(wild type yeast) and YGR079W(knockout yeast gene) growth after being subjected to 4mM caffeine solution. | ||
| + | |||
| + | [[Image:YGR079W.jpeg|500px]] | ||
| + | ===Interpretation=== | ||
| + | As seen in the growth chart, YGR079W has a much slower rate of growth when compared to the Wild-Type yeast. | ||
| + | |||
| + | ====Interpretation==== | ||
| + | |||
| + | ===[[UW-Stout/Hydrogen Peroxide SP22]]=== | ||
| + | |||
| + | As part of the University of Wisconsin Stout Orphan Gene Project this gene was tested by exposing the cells to hydrogen peroxide. | ||
| + | |||
| + | |||
| + | ====Results==== | ||
| + | |||
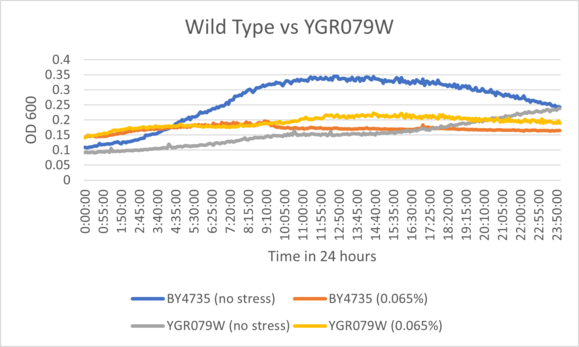
| + | BY4735 and YGR079W after being exposed to 0.065% dilution of hydrogen peroxide solution. | ||
| + | |||
| + | [[File:YGR079Wperosm.png]] | ||
| + | |||
| + | ====Interpretation==== | ||
| + | |||
| + | Based on the growth curve, YGR079W demonstrated close to standard inhibition under the stress of the hydrogen peroxide solution when compared to wild type cells. It's control exhibited slow growth but ended around the same value as wild type cells. | ||
| + | ===[[UW Stout/Nystatin SP22]]=== | ||
| + | As part of the University of Wisconsin Stout [[UW-Stout/About|Orphan Gene Project]] this gene was tested by exposing the cells to 1µg/ml Nystatin solution. | ||
| + | ====Results==== | ||
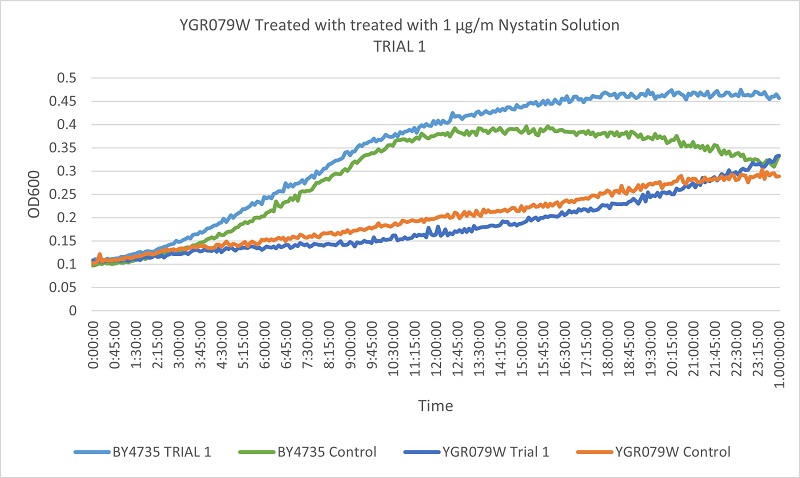
| + | [[File:YGR-T1.jpg]] | ||
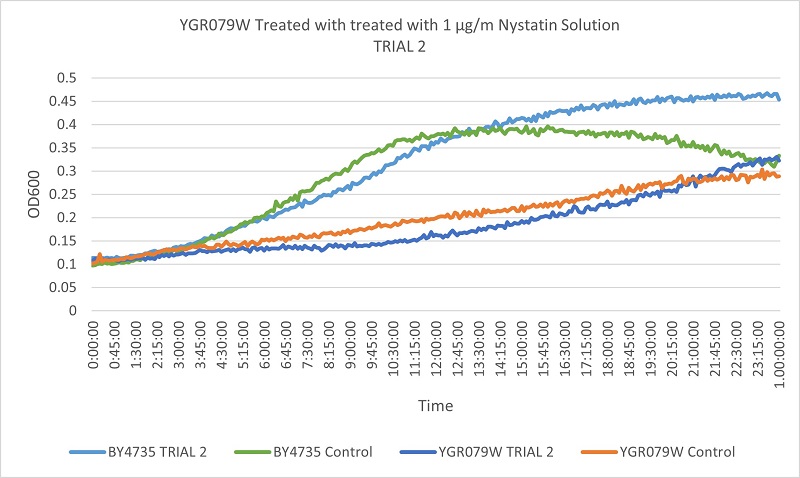
| + | [[File:YGR-T2.jpg]] | ||
| + | |||
| + | Based on the growth curve, we can see that the YGR079W strain is more slowly growing than the wild type yeast. The difference between the control and experimental in the wild type yeast and the YGR079W strain yeast is very similar. This indicates that the removal of the YGR079W gene has no effect on the cells resistance to Nystatin. | ||
| + | |||
| + | |||
| + | ===[[UW-Stout/Formamide SP22]]=== | ||
| + | |||
| + | |||
| + | |||
| + | ===Results=== | ||
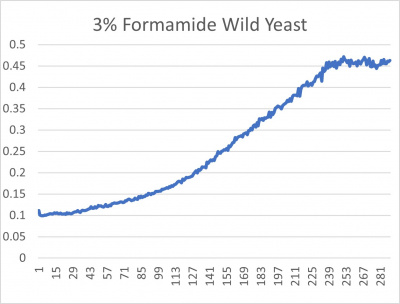
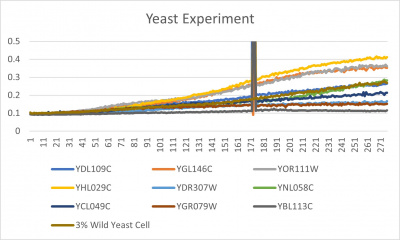
| + | [[File:wildyeast.jpg|400px|]] [[File:yeast.jpg|400px|]] [[File:doublingtimestable.jpg|400px|]] | ||
| + | |||
| + | |||
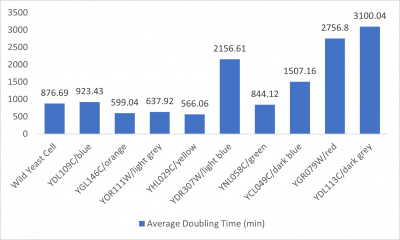
| + | *3% formamide line graph: x-axis= growing time (min); y-axis= optical density 600 | ||
| + | *Trial 2 line graph: x-axis= growing time (min); y-axis= optical density 600; key= yeast strains and corresponding color to the individual lines in the line graph | ||
| + | *Average doubling time bar graph: x-axis= yeast strains and corresponding colors to the trial 2 line graph; y-axis= average doubling time (min) | ||
| + | |||
| + | ===Interpretation=== | ||
| + | The wild yeast cell treated with 3% formamide had a doubling time of 876.69 min. The 3% formamide solution added to nine transformed yeast cells was then compared to the wild yeast cell using the computed doubling times to see the effects of the formamide. In the experiment, some of the transformed yeast cells were heavily effected by the formamide, including the YGR079W strain. The YGr079W strain had doubling times significantly slower than the wild yeast cell, with a doubling time of 2756.8 min. It can be concluded that the transformed yeast cell, YGR079W, had a unique response to the formamide stress test used in the experiment. | ||
| + | |||
==References== | ==References== | ||
<!-- REFERENCES ARE AUTOMATICALLY GENERATED. PLEASE DON'T EDIT THIS SECTION--> | <!-- REFERENCES ARE AUTOMATICALLY GENERATED. PLEASE DON'T EDIT THIS SECTION--> | ||
{{RefHelp}} | {{RefHelp}} | ||
</protect> | </protect> | ||
| + | |||
| + | '''[[UW-Stout/Nonionic Detergent SP22]]''' | ||
| + | |||
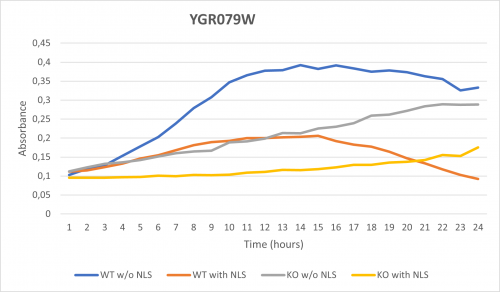
| + | As part of the University of Wisconsin Stout Orphan Gene Project this gene was tested by exposing the cells to 0.001% concentrated NLS. (Nonionic detergent) | ||
| + | |||
| + | [[File:YGR079W.png|500px]] | ||
| + | |||
| + | '''Interpretations:''' | ||
| + | |||
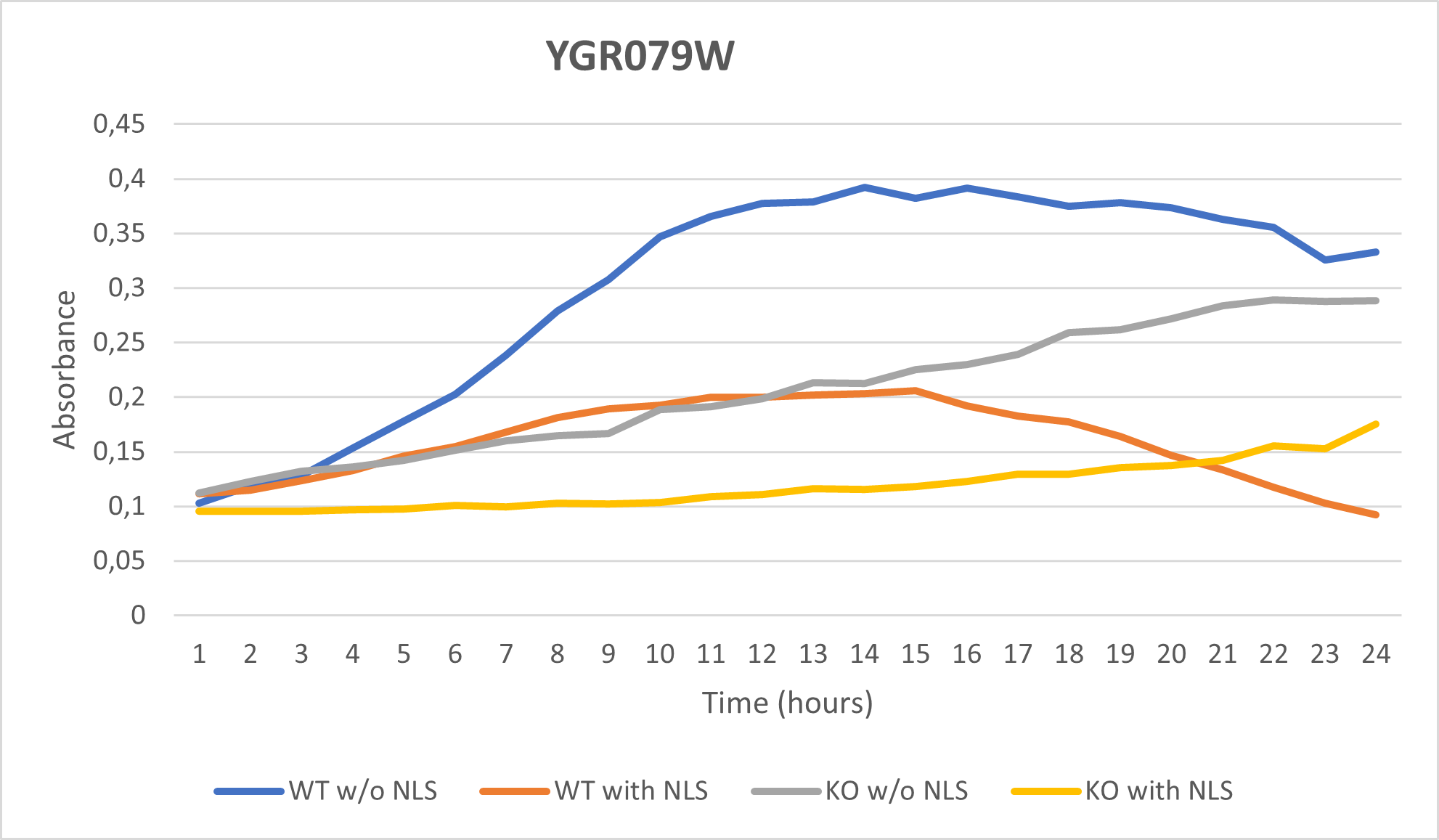
| + | -According to the graph above, we can see that the KO yeast cell YGR079W follow a smaller sensitivity to the stress than the Wild type strain. | ||
| + | |||
| + | -We can then conclude that the knocked-out gene in YGR079W doesn't have a significant impact on the growth of the yeast cell. | ||
| + | |||
| + | |||
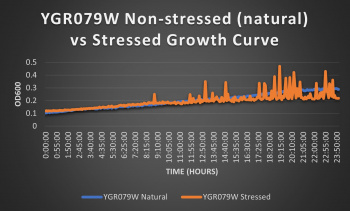
| + | '''[[UW-Stout/pH Acid SP22]]''' | ||
| + | |||
| + | [[File:Strain8.jpg|350px|]] | ||
| + | Knocked out gene seems to have no effect on cell growth in pH 7 environment based on the nearly identical natural and stressed growth curves. Spikes in stressed line are to be ignored; they occurred due to clumping of cells during analysis. We are focusing on the general linear trend of the growth curve. Please see protocol for specific quantitative doubling time results. | ||
| + | |||
| + | |||
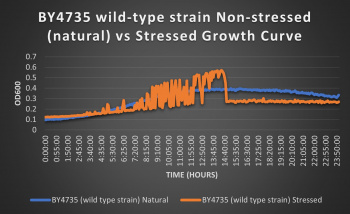
| + | [[File:Wild (1).jpg|350px|]] | ||
| + | The wild-type strain was used as a control in this experiment; no genes were knocked out, it just represents how the experiment ran on the knocked-out gene strains affects a "typical" cell growth curve. Inclusion of this data on each knock-out strain growth curve graph made for difficult interpretation, so it has been separated. Please refer to this graph as a control when viewing the knock-out strain growth curve graph. Spikes in stressed line are to be ignored; they occurred due to clumping of cells during analysis. We are focusing on the general linear trend of the growth curve. | ||
Latest revision as of 19:35, 11 May 2022
Share your knowledge...Edit this entry! <protect>
| Systematic name | YGR079W |
| Gene name | |
| Aliases | |
| Feature type | ORF, Uncharacterized |
| Coordinates | Chr VII:640720..641832 |
| Primary SGDID | S000003311 |
Description of YGR079W: Putative protein of unknown function; YGR079W is not an essential gene[1]
</protect>
Contents
- 1 Community Commentary
- 2 References
Community Commentary
About Community Commentary. Please share your knowledge!
DNA and RNA Details
Other DNA and RNA Details
Other Topic: expression
Specifically higher expression in phosphorus limited chemostat cultures versus phosphorus excess. [2] [3]
This gene is part of the UW-Stout Orphan Gene Project. Learn more here.
UW Stout/D2O SP22
The Knock Out Yeast Strain YGR079W (A8) Is more of a resistant strain to the 35% dilution of D20. With the range of maximum OD being about 0.2-0.3 OD 600. However, the growth of this is also a slower growth rate.
UW Stout/Sucrose Fermentation SP22
| Gene | Glucose | Fructose | Ethanol |
| Standard solution | 2.0000 | 0.2000 | 2.0000 |
| YDl109C | 0.3800 | 0.3933 | 0.3430 |
| YGL140C | 0.2212 | 0.2685 | 0.1867 |
| YOR111W | 0.3332 | 0.3598 | 0.1343 |
| YHL029C | 0.3870 | 0.2368 | 0.1151 |
| YDR307W | 0.4366 | 0.2487 | 0.0606 |
| YNL058C | 0.2710 | 0.3056 | 0.1577 |
| YCL049C | 0.4078 | 0.3052 | 0.1969 |
| YGR079W | 0.4042 | 0.1589 | 0.0080 |
| YBL113C | 0.3498 | 0.2012 | 0.1434 |
| BY4735 | 0.3171 | 0.3084 | 0.3541 |
Interpretation
This gene produced 2.26% of the amount of ethanol that the wild type produced. There is a high probability that this gene affects fermentation since there is such a low amount of ethanol produced but further tests are required.
<protect>
UW-Stout/UV Light SP22
As part of the University of Wisconsin Stout Orphan Gene Project this gene was tested under a UV Light using this protocol.
RESULTS
INTERPERTATION
In the graph and photos above, exposing this gene to 600 seconds of 400 Watt UV Light killed approximately 80% of yeast cell cultures, compared to its control counterpart, which was the same gene and amount of cells, just was not exposed to UV Light.
UW-Stout/Heat Shock SP22
As part of the University of Wisconsin Stout Orphan Gene Project this gene was tested by exposing the cells to heat shock.
Results
Interpretation
From the data gathered, there was a medium negative effect to knocking out this gene when it came to the yeast's ability to hold up to heat shock. The modified yeast cells were only 36% as resistant to heat shock than the control.
UW-Stout/Caffeine SP22
As part of the University of Wisconsin Stout Orphan Gene Project this gene was tested by exposing the cells to 4mM of caffeine following this protocol.
Results
- BY4735(wild type yeast) and YGR079W(knockout yeast gene) growth after being subjected to 4mM caffeine solution.
===Interpretation===
As seen in the growth chart, YGR079W has a much slower rate of growth when compared to the Wild-Type yeast.
Interpretation
UW-Stout/Hydrogen Peroxide SP22
As part of the University of Wisconsin Stout Orphan Gene Project this gene was tested by exposing the cells to hydrogen peroxide.
Results
BY4735 and YGR079W after being exposed to 0.065% dilution of hydrogen peroxide solution.
Interpretation
Based on the growth curve, YGR079W demonstrated close to standard inhibition under the stress of the hydrogen peroxide solution when compared to wild type cells. It's control exhibited slow growth but ended around the same value as wild type cells.
UW Stout/Nystatin SP22
As part of the University of Wisconsin Stout Orphan Gene Project this gene was tested by exposing the cells to 1µg/ml Nystatin solution.
Results
Based on the growth curve, we can see that the YGR079W strain is more slowly growing than the wild type yeast. The difference between the control and experimental in the wild type yeast and the YGR079W strain yeast is very similar. This indicates that the removal of the YGR079W gene has no effect on the cells resistance to Nystatin.
UW-Stout/Formamide SP22
Results
- 3% formamide line graph: x-axis= growing time (min); y-axis= optical density 600
- Trial 2 line graph: x-axis= growing time (min); y-axis= optical density 600; key= yeast strains and corresponding color to the individual lines in the line graph
- Average doubling time bar graph: x-axis= yeast strains and corresponding colors to the trial 2 line graph; y-axis= average doubling time (min)
Interpretation
The wild yeast cell treated with 3% formamide had a doubling time of 876.69 min. The 3% formamide solution added to nine transformed yeast cells was then compared to the wild yeast cell using the computed doubling times to see the effects of the formamide. In the experiment, some of the transformed yeast cells were heavily effected by the formamide, including the YGR079W strain. The YGr079W strain had doubling times significantly slower than the wild yeast cell, with a doubling time of 2756.8 min. It can be concluded that the transformed yeast cell, YGR079W, had a unique response to the formamide stress test used in the experiment.
References
See Help:References on how to add references
- ↑ Giaever G, et al. (2002) Functional profiling of the Saccharomyces cerevisiae genome. Nature 418(6896):387-91 SGD PMID 12140549
- ↑ Boer VM, et al. (2003) The genome-wide transcriptional responses of Saccharomyces cerevisiae grown on glucose in aerobic chemostat cultures limited for carbon, nitrogen, phosphorus, or sulfur. J Biol Chem 278(5):3265-74 SGD PMID 12414795
- ↑ submitted by Viktor Boer on 2003-07-25
See Help:Categories on how to add the wiki page for this gene to a Category </protect>
UW-Stout/Nonionic Detergent SP22
As part of the University of Wisconsin Stout Orphan Gene Project this gene was tested by exposing the cells to 0.001% concentrated NLS. (Nonionic detergent)
Interpretations:
-According to the graph above, we can see that the KO yeast cell YGR079W follow a smaller sensitivity to the stress than the Wild type strain.
-We can then conclude that the knocked-out gene in YGR079W doesn't have a significant impact on the growth of the yeast cell.
 Knocked out gene seems to have no effect on cell growth in pH 7 environment based on the nearly identical natural and stressed growth curves. Spikes in stressed line are to be ignored; they occurred due to clumping of cells during analysis. We are focusing on the general linear trend of the growth curve. Please see protocol for specific quantitative doubling time results.
Knocked out gene seems to have no effect on cell growth in pH 7 environment based on the nearly identical natural and stressed growth curves. Spikes in stressed line are to be ignored; they occurred due to clumping of cells during analysis. We are focusing on the general linear trend of the growth curve. Please see protocol for specific quantitative doubling time results.
 The wild-type strain was used as a control in this experiment; no genes were knocked out, it just represents how the experiment ran on the knocked-out gene strains affects a "typical" cell growth curve. Inclusion of this data on each knock-out strain growth curve graph made for difficult interpretation, so it has been separated. Please refer to this graph as a control when viewing the knock-out strain growth curve graph. Spikes in stressed line are to be ignored; they occurred due to clumping of cells during analysis. We are focusing on the general linear trend of the growth curve.
The wild-type strain was used as a control in this experiment; no genes were knocked out, it just represents how the experiment ran on the knocked-out gene strains affects a "typical" cell growth curve. Inclusion of this data on each knock-out strain growth curve graph made for difficult interpretation, so it has been separated. Please refer to this graph as a control when viewing the knock-out strain growth curve graph. Spikes in stressed line are to be ignored; they occurred due to clumping of cells during analysis. We are focusing on the general linear trend of the growth curve.