UW Stout/D2O SP22
Contents
Introduction
D2O also known as Heavy Water is a form of water that is deuterium. It is heavier than normal H2O and can have many physical and chemical properties that are different than normal H2O. It is known to slow down neutrons and, in some cases, possibly absorb these neutrons. Although it is similar to water it can only be somewhat safely consumed in very small quantities. The experimentation process uses a Wild-Type yeast cell as a control and 9 Knockout Strains for the experimentation. With this, we will observe and record the growth curves of these cells using Optical Density 600 in a 24-hour observation period which in return will tell us the number of cells in each well plate.
Materials and Amounts
- 1ml D2O
- Sterile Water
- Wild-type yeast in 2x synthetic complete media at an OD600 of 0.1-0.2
- Corning COSTAR 96-well clear flat-bottom assay plate
- 9 Knockout Yeast Strains
Equipment
- Biosafety Cabinet
- Micro-pipetting tips
- Micropipettes
- Molecular Devices Spectra Max Plus 384 Microplate Reader
Safety
- Safety Glasses
- Lab Jacket
- Gloves
- Safety Data Sheet for D2O
Procedure
- Diluting the D2O with sterile water, by following the dilution rates below
- 50% dilutions (25ul D2O to 25ul H2O) (10 samples of this dilution)(Used only in Pilot)
- 0% dilutions (50ul H2O) (10 samples of this dilution)(Control)
- 75% dilutions (37.5ul D2O with 12.5 ul of H2O)(Used only in Pilot)
- 35% Dilutions (17.5ul D2O with 32.5ul of H2O)(Final Dilution)
- 20% dilutions (10ul of D2O with 40ul of H2O)(Used only in Pilot)
- Vortex the yeast culture briefly to resuspend the yeast cells.
- Set up the plates as 50ul of dilution a with 50ul of Yeast cells (Well 1-9) and dilution as 50ul dilution and 50ul H2O (Well 10)
- Set up the plate reader as follows:
- Temperature: 30°C
- Mode: Kinetic
- Wavelength: 600 nm
- Interval: 5 minutes
- Total run time: 24 hours
- Shake before reading: 30 seconds
- Transfer the assay plate to the reader and read for 24 hours.
Pilot Experiment with Wild Type Yeast Cells
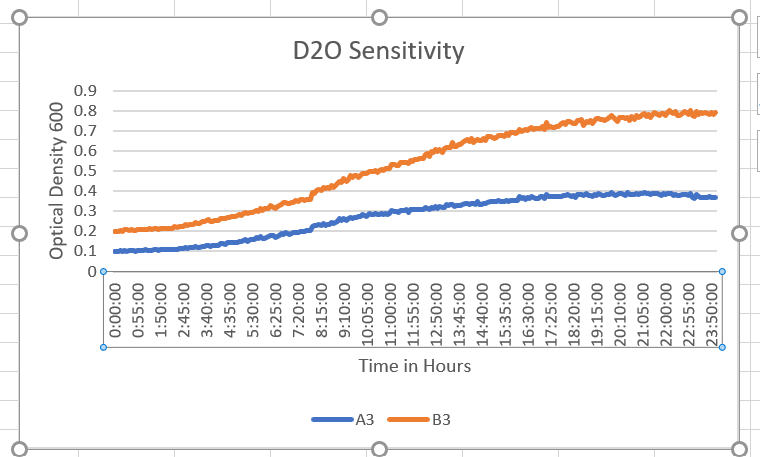
- On 4/12/2022 we did a pilot experimentation process with a 50% dilution and control. They showed that the OD 600, had good results (inserted below). They showed an increase in the OD 600 and a turn in the number of cells in the well of the plate compared to the control dilution. This is telling us that cells are growing less in the D2O diluted with water rather than in the pure water control (B3).B3- Control A3-50% dilution. As you can see the control grew at a higher rate than the 50% D2O dilution. Th control hit about 0.8 whereas the dilution hit about 0.4. This gave us a great start to see the D2O is decreasing the growth rate of the yeast cells.
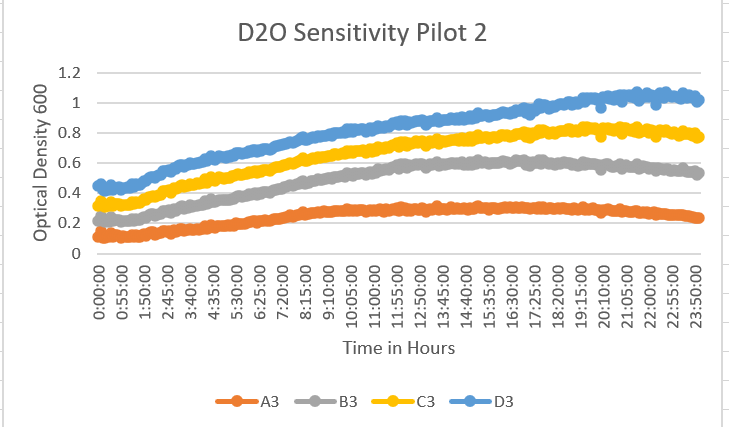
- We ran a pilot experiment with a control of 75%, 35%, and 20%. This was to fill in the gray area of our last pilot experiment and this led us to see some great information and data to help us with our overall experiment. A3 was 75% dilution. B3 was 35% dilution, C3 was 20% dilutions, and D3 was the control at 0% dilution. This showed again an increase in the OD 600 and that the number of cells in the well decreased. It shows that as the dilution increased so did the OD 600 of each dilution and it perfectly fills in the areas of the previous graph. As you can see the OD 600 decreases and then seems to plateau during the 24 hours. With the higher, concentration of the dilution came a lower OD 600 value. As you can see the 75% dilution hit about 0.15, the 35% dilution hit about 0.6, the 20% dilution hit about 0.8 and the control hit 1.0. This data helped us to select the 35% dilution by how much the D2O dilution decreased the growth rate.
Final Experimentation with 35% D2O Dilution and 9 Knockout-Yeast Strains
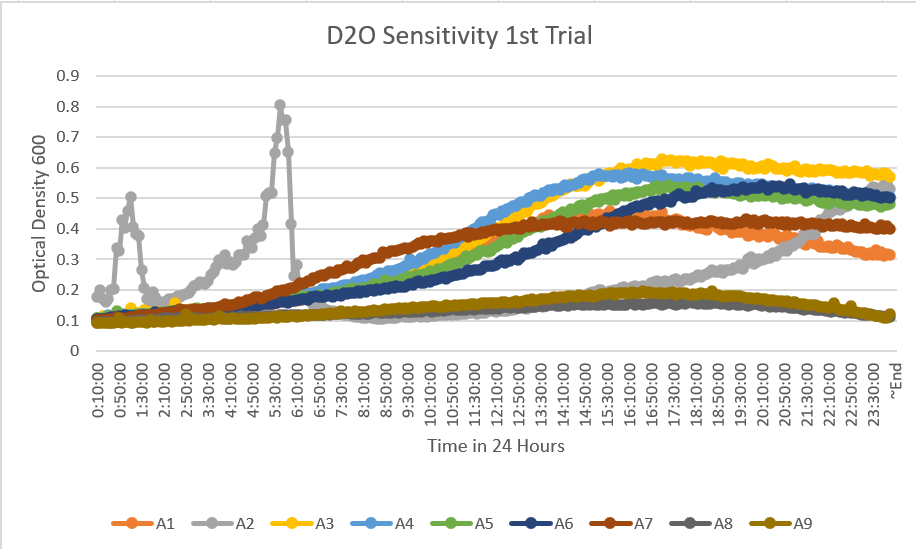
- On Tuesday we completed the first set of trials however we will use this as a pilot due to a mistake made between the plating. We added yeast 1 and 2 together on accident, and 8 and 9 we did not dilute properly according to are calculations due to an error on our part. So, although we have this data, we will be doing this first run-through again in the hope of getting more accurate to the actual reaction and their rates with the D20 dilution and yeast knockout strains. A1 control. A2- YDL109C, A3- YGL146C, A4- YOR111W, A5- YHL029C, A6- YDR307W, A7- YNL058C, A8- YCL049C, A9- YGR079W, A10- YBL113C
First Successful Experimentation
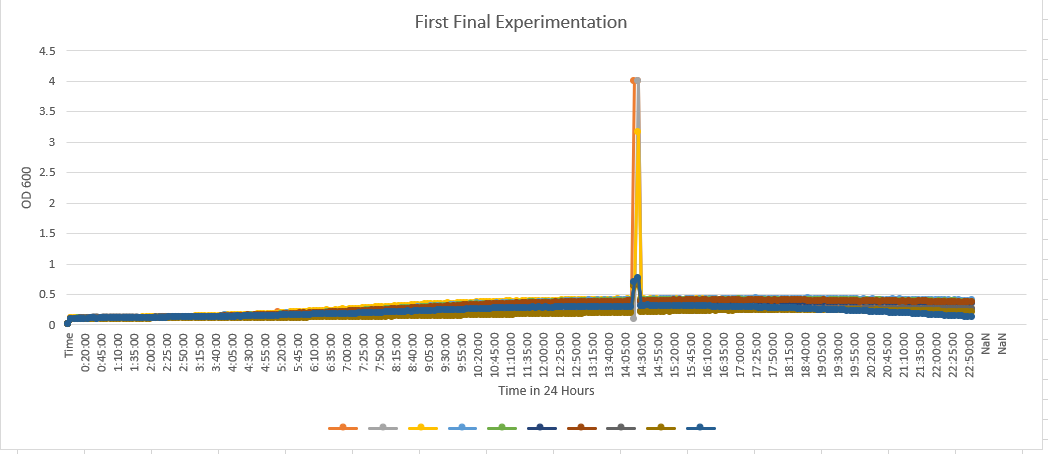
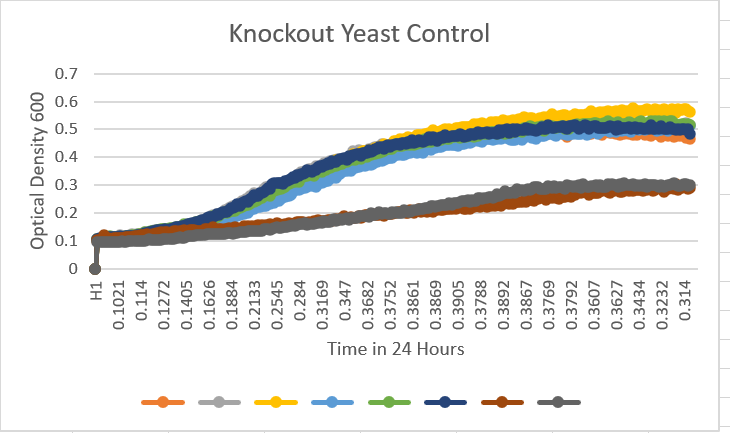
- A large piece to note with this data is that there are some outliers that Dr.Teague informed us to ignore in the overall results. These stated outliers skewed the overall view of the graph to be noted. A10 is the control medium blue). A1 with YDL109C(orange), A2 with YGL146C(gray), A3 with YOR111W(yellow), A4 with YHL029C(light blue), A5 with YDR307W(green), A6 with YNL058C(dark blue), A7 with YCL049C(deep red), A8 with YGR079W(dark gray), A9 with YBL113C(brown). The data matches very well with the knockout verification controls. They have very similar growth curves. With the YDL109C reaching about 0.46 OD, YGL146C reaching about 0.26, YOR111W reaching about 0.48, YHL029C reaching about 0.48, YDR307W reaching 0.47, YNL058C reaching around 0.30, YCL049C reaching near 0.45, YGR079W reaching 0.20, YBl113C reaching 0.15, and finally the control with the wild type yeast cell reaching around, the midline at about 0.26. With the controls strains being so similar it tells us that D2O has an affect but not too much of an affect that it kills it off or completely stops the growth.
- Compared to
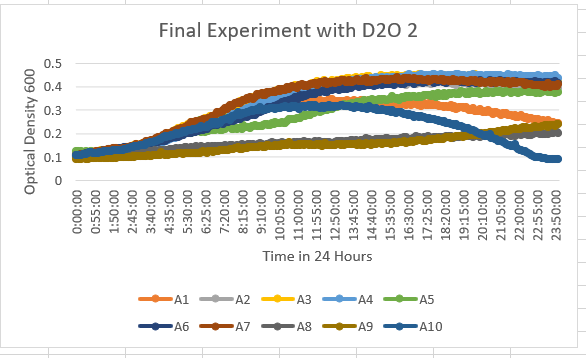
Second Successful Experimentation
- A1 with YDL109C(orange), A2 with YGL146C(gray), A3 with YOR111W(yellow), A4 with YHL029C (light blue), A5 with YDR307W(green), A6 with YNL058C (dark blue), A7 with YCL049C(deep red), A8 with YGR079W(dark gray), A9 with YBL113C(brown). Now that we do not have an outlier present, we can see a better ratio of how the results came out. With this, you can notice that all of them have a similar OD of 600 within a 0.1 to about 0.47 OD ratio between them all. They all start at the same point with them increasing to hit more of a plateau at about 10 hours. With this data, we can confirm that the D2O in influencing the yeast strains that we tested.
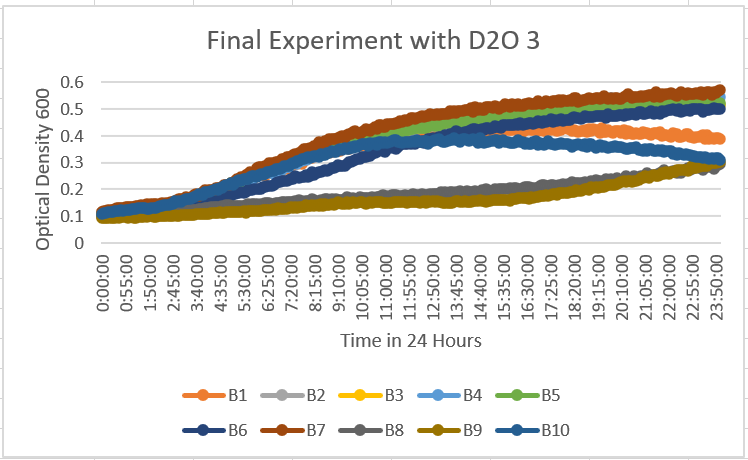
Third Successful Experimentation
- B1 with YDL109C(orange), B2 with YGL146C(gray), B3 with YOR111W(yellow), B4 with YHL029C (light blue), B5 with YDR307W(green), B6 with YNL058C (dark blue), B7 with YCL049C (deep red), B8 with YGR079W (dark gray), B9 with YBL113C(brown). With this data, we see some more confirming results from the 1st and 2nd experimentations and their results. However, we can see the B9 with YBL113C(brown) and), B8 with YGR079W (dark gray). They seem to be having a different type of reaction than the others and their past results. We can also see that the range ratio this time is 0.1-0.58 instead of below 0.5 OD 600. This may be telling us there may have been a small mistake or just a variation in the reaction with these yeast strains and to 35% D2O solution.