Difference between revisions of "UW-Stout/Copper FA21"
| Line 44: | Line 44: | ||
'''Part 2''' | '''Part 2''' | ||
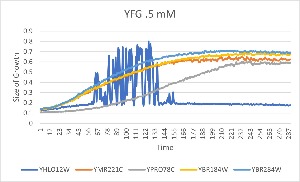
[[File:YFG1.jpg]] | [[File:YFG1.jpg]] | ||
| − | In the above graph, you can see that there is an anomaly in the data that at first we assumed was either a pipetting or instrument error. However, when running the experiment again we got the same result and we believe the growth is drying up. | + | :In the above graph, you can see that there is an anomaly in the data that at first we assumed was either a pipetting or instrument error. However, when running the experiment again we got the same result and we believe the growth is drying up. |
#Finally we did the same process but in the 2 rows of wells surrounding our yeast we put 100ul of distilled water to try to avoid evaporation of our yeast to try to avoid the same error happening a third time | #Finally we did the same process but in the 2 rows of wells surrounding our yeast we put 100ul of distilled water to try to avoid evaporation of our yeast to try to avoid the same error happening a third time | ||
Revision as of 09:37, 15 December 2021
Contents
Materials & Equipment
- CuSO4
- Sterile Water
- 1000ul Pipette
- 50ul Pipette
- Centrifuge Tubes
- Wild Type Yeast Cells
- Yeast Cells with Your Favorite Gene removed.
- Vortex
- Disposable Pipette Tips
- Plate Reader
- Wells
Personal Protective Equipment
- Latex Gloves
- Eye Pro
- Lab Coat
Wild Type Protocol
- Vortex Wild type Yeast Solutions
- Create Solutions with Copper Sulfate and Sterile water at the following Concentrations.
- 20mM (Solution) Copper Sulfate
- 10mM Copper Sulfate
- 5.0mM Copper Sulfate
- 2.0mM Copper Sulfate
- 1.0mM Copper Sulfate
- 0.5mM Copper Sulfate
- 0.2mM Copper Sulfate
- 0.1mM Copper Sulfate
- Combine 50ul of the Copper Sulfate solution and 50ul of the wild type yeast cells and put them in eight different wells.
- Take the wells that are prepared with the yeast and the Copper Sulfate and put it in a plate reader and leave it there for 24 hours.
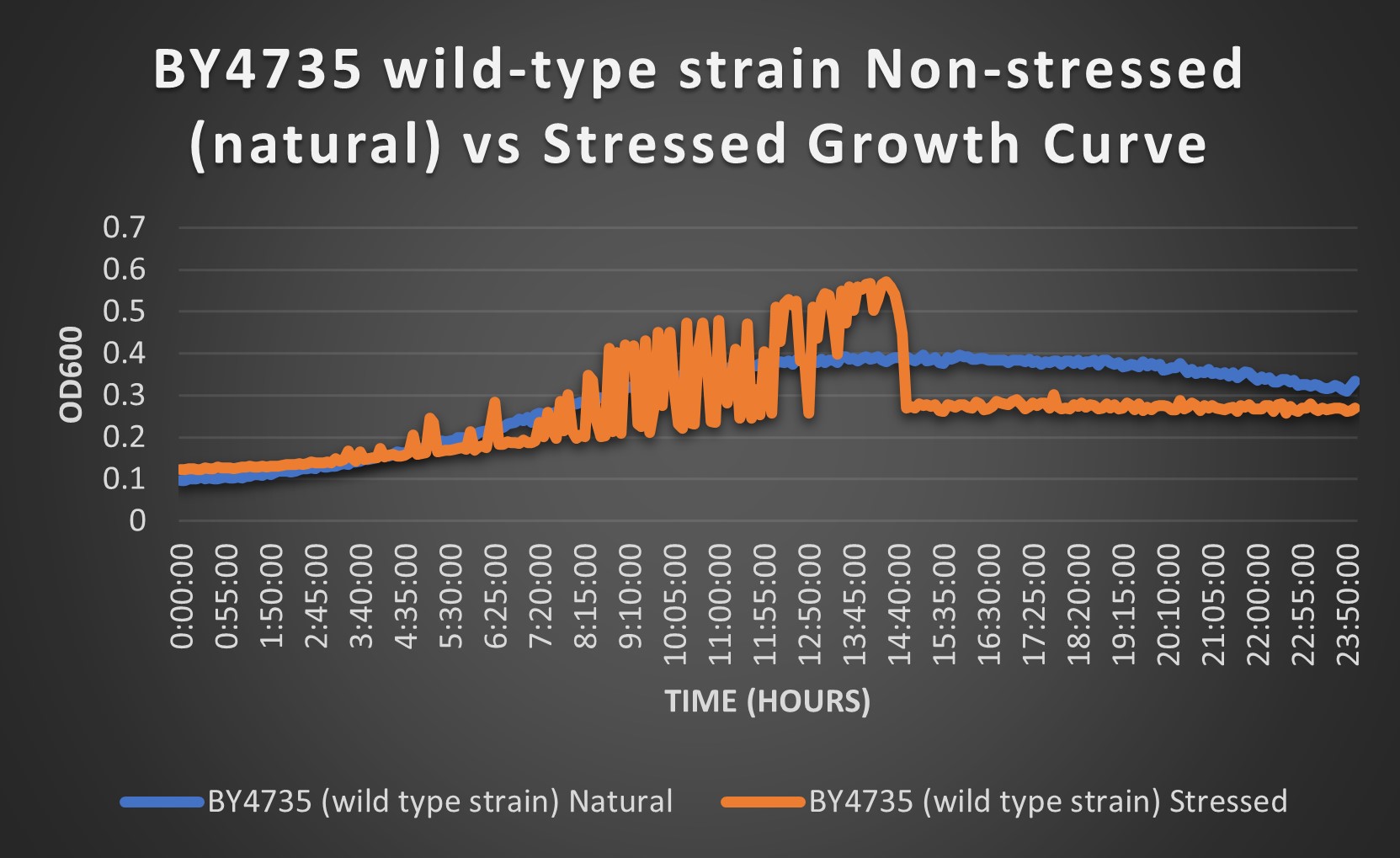
Interpreting Wild Type Data
Protocol with Gene Removed
Part 1
- Insert 50ul of yeast cells in different wells.
- Insert 50ul of CuSO4 at 0.5mM concentration into the same wells as the yeast cells.
- Interpret the data on a plate reader.
- Graph collected data and compute average doubling time for each yeast strain.
- Repeat this process again and analyze the data.
- In the above graph, you can see that there is an anomaly in the data that at first we assumed was either a pipetting or instrument error. However, when running the experiment again we got the same result and we believe the growth is drying up.
- Finally we did the same process but in the 2 rows of wells surrounding our yeast we put 100ul of distilled water to try to avoid evaporation of our yeast to try to avoid the same error happening a third time