Difference between revisions of "UW-Stout/G418 FA21"
(→Calibration Protocol) |
(→KO Gene Protocol) |
||
| Line 331: | Line 331: | ||
'''Data''' | '''Data''' | ||
| − | [[File: | + | [[File:bettergraph2.png]] |
Analysis of our data through doubling rates: | Analysis of our data through doubling rates: | ||
[[File:Data_Ananysis_-_Yeast_KO_Genes.xlsx]] | [[File:Data_Ananysis_-_Yeast_KO_Genes.xlsx]] | ||
Latest revision as of 14:04, 20 December 2021
Calibration Protocol
Materials
Safety Gloves
Safety Glasses
Well Plate
Molecular Devices SpectraMax Plus 384 Microplate Reader
Fume Hood
Sterile Water
G418
Pipettes
Wild Type Yeast
5 Knockout Yeast
Protocol
1. Obtain 10 different concentrations of the G418 antibiotic and sterile water. Make sure to have 1 extra concentration of sterile water as a control growth curve of the wild type yeast.
2. Set up 11 wells shown in the "Well Components".
3. Put the well plate to the reader and read for 24 hours.
4. Observe the the growth curves.
5. Determine which concentration should be used on the knockout yeast.
Concentrations Used
1. 100mg/ml G418
- 200ul G418
-0ul water
2. 33.3mg/ml G418
-66.7ul G418
-133.3ul water
3. 11.1mg/ml G418
-22.2ul G418
-177.8ul water
4. 3.7mg/ml G418
-7.4ul G418
-192.6ul water
5. 1.23mg/ml G418
-2.5ul G418
-197.5ul water
6. 0.41mg/ml G418
-1.6ul G418
-398.4ul G418
7. 0.41mg/ml G418
-1.1ul G418
-800ul water
8. 0.0456mg/ml G418
-1.4ul G418
-3,000ul water
9. 0.0152mg/ml G418
-1.5ul G418
-10,000ul water
10. 0.0050mg/ml G418
-1.0ul G418
-20,000ul water
11. 0mg/ml G418 (control)
-0ul G418
-200ul water
Note: For concentrations 6-10, serial dilutions were made. This is because the amount of G418 used would have been to small to physically preform.
Well Components
Well 1:
-50 ul wt yeast
-50 ul 100mg/ml G418
Well 2:
-50 ul wt yeast
-50 ul 33.3mg/ml G418
Well 3:
-50 ul wt yeast
-50 ul 11.1mg/ml G418
Well 4:
-50 ul wt yeast
-50 ul 3.7mg/ml G418
Well 5:
-50 ul wt yeast
-50 ul 1.23mg/ml G418
Well 6:
-50 ul wt yeast
-50 ul 0.41mg/ml G418
Well 7:
-50 ul wt yeast
-50 ul 0.137mg/ml G418
Well 8:
-50 ul wt yeast
-50 ul 0.0456mg/ml G418
Well 9:
-50 ul wt yeast
-50 ul 0.0152mg/ml G418
Well 10:
-50 ul wt yeast
-50 ul 0.0050mg/ml G418
Well 11:
-50 ul wt yeast
-50 ul water
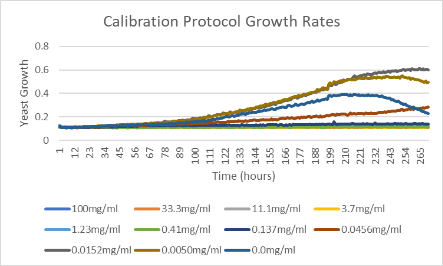
Data
Based on the data from the Molecular Devices SpectraMax Plus 384 Microplate Reader, we decided to use the concentration from well 8 (E8).
KO Gene Protocol
Materials
Safety Gloves
Safety Glasses
Well Plate
Molecular Devices SpectraMax Plus 384 Microplate Reader
Fume Hood
Sterile Water
G418
Pipettes
Wild Type Yeast
5 Knockout Yeast
Protocol
1. Obtain the concentration determined from the calibration protocol (0.0456mg/ml G418).
2. Set up 12 wells as described in "Well Components".
3. In the well plates there will be 2 of the same components. (There are 5 KO yeasts and 1 wild type)
4. Put the well plate to the reader and read for 24 hours.
5. Observe the the growth curves.
Well Components
Well 1:
-50 ul wt yeast
-50 ul 0.0456mg/ml G418
Well 2:
-50 ul YHL012W gene
-50 ul 0.0456mg/ml G418
Well 3:
-50 ul YMR221C gene
-50 ul 0.0456mg/ml G418
Well 4:
-50 ul YPR078C gene
-50 ul 0.0456mg/ml G418
Well 5:
-50 ul YBR184W gene
-50 ul 0.0456mg/ml G418
Well 6:
-50 ul YBR284W gene
-50 ul 0.0456mg/ml G418
Well 7:
-50 ul wt yeast
-50 ul 0.0456mg/ml G418
Well 8:
-50 ul YHL012W gene
-50 ul 0.0456mg/ml G418
Well 9:
-50 ul YMR221C gene
-50 ul 0.0456mg/ml G418
Well 10:
-50 ul YPR078C gene
-50 ul 0.0456mg/ml G418
Well 11:
-50 ul YBR184W gene
-50 ul 0.0456mg/ml G418
Well 12:
-50 ul YBR284W gene
-50 ul 0.0456mg/ml G418
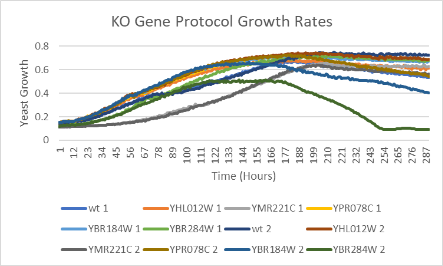
Data
Analysis of our data through doubling rates: